resonance (in chemistry)
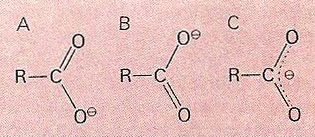
A carboxylate ion can be thought of as a resonance hybrid between valence bond structures (A) and (B). Alternatively, the hybrid structure can be represented as (C)
In chemistry, resonance refers to the theory of molecule structure in which the actual state of the bonding in a molecule is expressed as a 'resonance hybrid' between two or more valence-bond structures, and is intermediate between them, but of lower energy. There is no actual oscillation, and the model is equivalent to molecular orbital theory. First proposed for benzene, resonance stabilizes aromatic compounds and conjugated double-bonded systems such as 1,3-butadiene.


