alkaline fuel cell
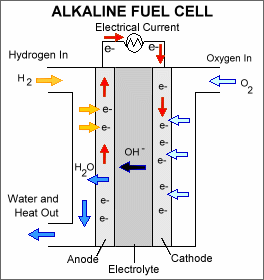
An alkaline fuel cell consists of an alkaline electrolyte, typically potassium hydroxide (KOH), sandwiched between an anode (negatively charged electrode) and a cathode (positively charged electrode). The processes that take place in the fuel cell are as follows: 1. Hydrogen fuel is channeled through field flow plates to the anode on one side of the fuel cell, while oxygen from the air is channeled to the cathode on the other side of the cell. 2. At the anode, a platinum catalyst causes the hydrogen to split into positive hydrogen ions (protons) and negatively charged electrons. 3. The positively charged hydrogen ions react with hydroxyl (OH-) ions in the electrolyte to form water. 4. The negatively charged electrons cannot flow through the electrolyte to reach the positively charged cathode, so they must flow through an external circuit, forming an electrical current. 5. At the cathode, the electrons combine with oxygen and water to form the hydroxyl ions that move across the electrolyte toward the anode to continue the process.
Alkaline fuel cells (AFCs) were one of the first fuel cell technologies developed, and they were the first type widely used in the U.S. space program to produce electrical energy and water on-board spacecrafts. These fuel cells use a solution of potassium hydroxide in water as the electrolyte and can use a variety of non-precious metals as a catalyst at the anode and cathode. High-temperature AFCs operate at temperatures between 100°C and 250°C (212°F and 482°F). However, newer AFC designs operate at lower temperatures of roughly 23°C to 70°C (74°F to 158°F).
AFCs' high performance is due to the rate at which chemical reactions take place in the cell. They have also demonstrated efficiencies near 60% in space applications.
The disadvantage of this fuel cell type is that it is easily poisoned by carbon dioxide (CO2). In fact, even the small amount of CO2 in the air can affect this cell's operation, making it necessary to purify both the hydrogen and oxygen used in the cell. This purification process is costly. Susceptibility to poisoning also affects the cell's lifetime (the amount of time before it must be replaced), further adding to cost.
Cost is less of a factor for remote locations, such as space or under the sea. However, to effectively compete in most mainstream commercial markets, these fuel cells will have to become more cost-effective. AFC stacks have been shown to maintain sufficiently stable operation for more than 8,000 operating hours. To be economically viable in large-scale utility applications, these fuel cells need to reach operating times exceeding 40,000 hours, something that has not yet been achieved due to material durability issues. This obstacle is possibly the most significant in commercializing this fuel cell technology.
