solid oxide fuel cell
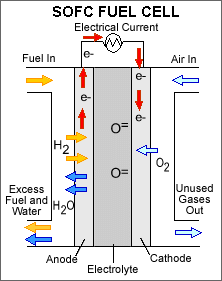
A solid oxide fuel cell consists of a non-porous metal oxide electrolyte (typically zirconium oxide) sandwiched between an anode (negatively charged electrode) and a cathode (positively charged electrode). The processes that take place in the fuel cell are as follows: 1. Hydrogen fuel is channeled through field flow plates to the anode on one side of the fuel cell, while oxygen from the air is channeled to the cathode on the other side of the cell. 2. At the cathode, a catalyst causes electrons from the electrical circuit to combine with oxygen to create negatively charged oxygen ions. 3. The negatively charged oxygen ions flow through the electrolyte to the anode. 4. At the anode, the catalyst causes the hydrogen to react with the oxygen ions forming water and free electrons. 5. The negatively charged electrons cannot flow through the electrolyte to reach the positively charged cathode, so they must flow through an external circuit, forming an electrical current. 6. At the cathode, the electrons combine with oxygen to create negatively charged oxygen ions, and the process repeats.
Solid oxide fuel cells (SOFCs) use a hard, non-porous ceramic compound as the electrolyte. Because the electrolyte is a solid, the cells do not have to be constructed in the plate-like configuration typical of other fuel cells types. SOFCs are expected to be around 50%–60% efficient at converting fuel to electricity. In applications designed to capture and utilize the system's waste heat (co-generation), overall fuel use efficiencies could top 80%–85%.
Solid oxide fuel cells operate at very high temperatures – around 1,000°C (1,830°F). High-temperature operation removes the need for precious-metal catalyst, thereby reducing cost. It also allows SOFCs to reform fuels internally, which enables the use of a variety of fuels and reduces the cost associated with adding a reformer to the system.
SOFCs are also the most sulfur-resistant fuel cell type; they can tolerate several orders of magnitude more of sulfur than other cell types. In addition, they are not poisoned by carbon monoxide (CO), which can even be used as fuel. This property allows SOFCs to use gases made from coal.
High-temperature operation has disadvantages. It results in a slow startup and requires significant thermal shielding to retain heat and protect personnel, which may be acceptable for utility applications but not for transportation and small portable applications. The high operating temperatures also place stringent durability requirements on materials. The development of low-cost materials with high durability at cell operating temperatures is the key technical challenge facing this technology.
Scientists are currently exploring the potential for developing lower-temperature SOFCs operating at or below 800°C that have fewer durability problems and cost less. Lower-temperature SOFCs produce less electrical power, however, and stack materials that will function in this lower temperature range have not been identified.
