mass spectrometry
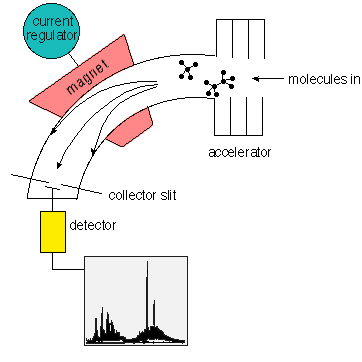
Mass spectrometry is an analytical technique in which ions are separated according to the mass/charge (m/e) ratio and detected by a suitable detector. In a mass spectrometer a sample is ionized and the positive ions produced are accelerated into a high-vacuum region containing electric and magnetic fields. These fields deflect and focus the ions onto a detector. A mass spectrum is thus obtained consisting of a series of peaks of variable intensity to which m/e values can be assigned. Different molecules can be identified by their characteristic pattern of lines.
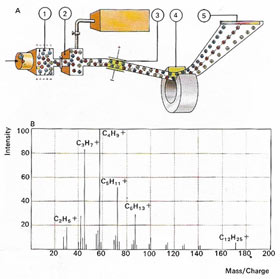 |
| In a mass spectrometer (A), outer electrons of a compound are removed in an ionization camber (1). Positively charged ions pass into an adjacent chamber under vacuum (2) and are focused by electric (3) and magnetic (4) fields. The way they are deflected by these fields is characteristic for each ion, which can be identified by its position on a photographic plate (5). A molecule such as n-dodecane breaks down into a number of fragments that produce various "peaks" on a graph (B). From the position of these, the parent molecules can be precisely identified.
|


