oxalic acid
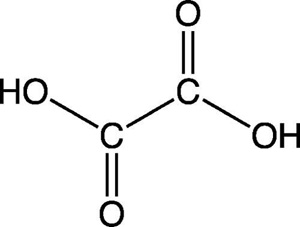
Oxalic acid is a white, crystalline solid, a dicarboxylic acid (dibasic carboxylic acid) that is slightly soluble in water. It is also known by the systematic name ethanedioic acid and has the chemical formula (COOH)2, or C2H2O4.
Oxalic acid is very poisonous and occurs in certain plants such as sorrel and the leaf blades of rhubarb. It is a mild reducing agent and is used as a standard in volumetric analysis. It is also used for metal and textile cleaning, and in tanning. Molecular weight 90.0, relative density 1.653, melting point 101.5°C (214.7°F).
Oxalic acid poisoning
Oxalic acid is a powerful local irritant; when swallowed it produces a burning sensation in the mouth and throat, vomiting of blood, breathing difficulties, and circulatory collapse. Treatment is with calcium lactate or other calcium salts, lime water, or milk.


