standard solution
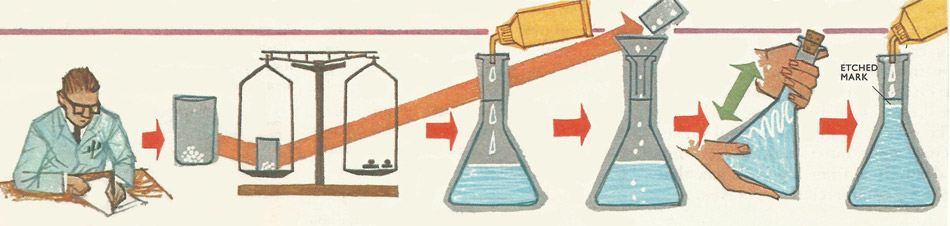
Preparing a standard solution.
A standard solution is a solution whose strength is accurately known. It can be used to find the strength of other solutions with which it reacts. Volumetric analysis is generally carried out with decinormal (0.1 N) solutions.
The first stage in making a standard solution is to work out the gram equivalent weight of the substance. To do this the gram molecular weight is found by adding up the atomic weights of the various atoms in the molecule. The reactions in which the substance takes part will indicate the relationship between the gram equivalent weight and the gram molecular weight.
If a decliner solution is to be made, one tenth of the gram equivalent of the substance is weighed out into a tared weighing bottle. A clean one-liter measuring flask is then partly filled with distilled water. The crystals of the substance are then carefully transferred to the flask (without losing any on the way). The flask is then shaken to dissolve the crystals which should be fairly small in size. When they have all dissolved, more distilled water is added until the liquid level corresponds with the one mark etched in the neck of the flask.
It is not possible to make up standard solutions of all substances in this way. For instance, concentrated sulfuric acid almost certainly contains some water, while pellets of sodium hydroxide actually absorb water while they are being weighed. So to obtain standard solutions of these substances, solutions of about the correct strength are prepared. The exact strength is then found by titration with a solution whose strength is accurately known.


