EDTA
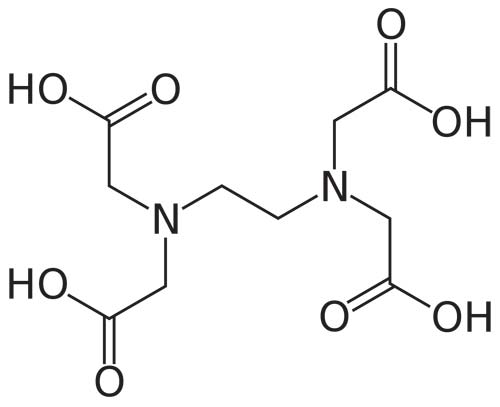
Skeletal formula of ethylenediaminetetraacetic acid.
EDTA, or ethylenediaminetetraacetic acid (HOOCCH2)2N(CH2)2N(CH2COOH)2), also called diaminoethane tetracarboxylic acid, is a chelating agent (see chelation). The acid binds with calcium, magnesium, and iron, and is used in chemical analysis, water softening, and to counteract metallic poisons that have been swallowed. In such applications EDTA actsas a sequestering agent.


