equilibrium

Figure 1. A body is in translational equilibrium when it is motionless and all the physical forces acting on it are mutually balanced. Such conditions are associated with peaks or wells in the body's potential-energy curve. A brick standing on its end (A) is said to be in a position of metastable equilibrium because, although it occupies a well in the potential-energy curve, it is not the deepest well available: by adding to the body's potential energy it can be pushed through a position of unstable equilibrium (B – a peak in the energy curve), whence it call fall to the position of stable equilibrium (C – the deepest well in the potential-equilibrium curve). A further position, called neutral equilibrium, is exemplified by a sphere resting on a horizontal plane: no horizontal displacement can alter its potential energy.

Figure 2. The very low center of gravity of the bus keeps it in stable equilibrium even for large displacements.
Equilibrium is a state in which a mechanical, thermodynamic, electrical, or other system will remain if undisturbed (Figure 1). In stable equilibrium the system returns to its original position if disturbed (Figure 2); the position of stable equilibrium thus determines the rest position of the system. In unstable equilibrium (as with a tall pole balanced on one end) it moves farther away. Stable and unstable equilibria correspond to configurations with minimal and maximal energy respectively.
A metastable state is an uneasy stability of a system that can easily be disturbed, making it descend to a lower energy state (a coin balanced on its edge is an example). A supersaturated solution or supercooled liquid, which remains liquid below its freezing point, is also metastable because the addition of a speck of dust or a tiny seed crystal will make it rapidly crystallize.
Mechanical equilibrium is a stable state in which forces acting on a particle or object cancel each other out, resulting in no net force. While thought of as a state of balance or rest, an object with constant velocity is also said to be in equilibrium.
A system is defined to be in thermodynamic equilibrium when it has the maximum possible amount of entropy.
The concept originated in ancient Greece when Archimedes experimented with levers in balance, literally "equilibrium." The idea was elaborated through the Middle Ages, the Renaissance, and the birth of modern mathematics and physics in the seventeenth and eighteenth centuries. In general parlance "equilibrium" has come to mean pretty much the same thing as stability, i.e., a system that is largely unaffected by internal or external changes since it easily returns to its original condition after being perturbed.
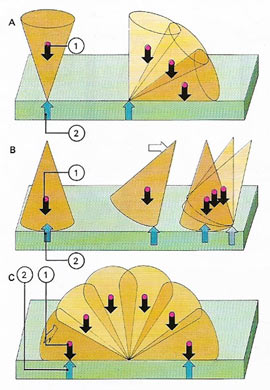 |
| If a cone standing on its point [A] is slightly displaced, its weight produces a moment that continues to topple the cone about this point of contact. This is an unstable equilibrium position. With the cone on its base [B], displacement produces a moment that restores the original position – a stable equilibrium. If the cone lies on its side [C], displacement produces no moment since the weight and its reaction [1,2] still act along the same line – the cone remains in its new position and is in neutral equilibrium.
|


