ester
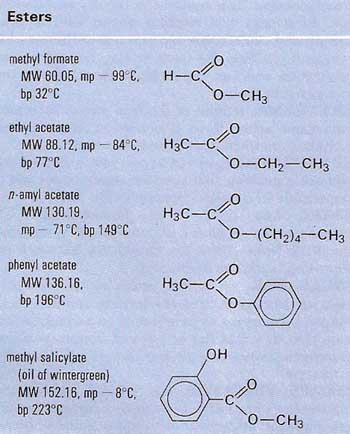
An ester is an organic compound formed by condensation of an acid (organic or inorganic) and an alcohol. This reaction, called esterification, is acid-catalyzed; its reverse, hydrolysis, is acid or base-catalyzed; an equilibrium is set up in aqueous solution. Esters formed from carboxylic acids have the general formula RCOOR'. Examples include ethyl ethanoate (CH3COOC2H5) and methyl propanoate (C2H5COOCH3).
Esters containing simple hydrocarbon groups are volatile, fragrant substances used as flavorings in the food industry. Triesters, molecules containing three ester groups, occur in nature as oils and fats (see triglyceride).
Polyester
A polyester is any of a class of organic substances composed of large molecules arranged in either a chain or a network and formed from many smaller molecules through the establishment of ester linkages. Polyester fibers are resistant to chemicals and may be washed in alkaline solutions or dry-cleaned. They are made into woven and knitted fabrics. As a clothing material, polyester is valued for its resistance to creasing.


