buckyball
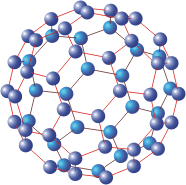
A buckyball, also known as buckminsterfullerene or fullerene, is a large molecule made of carbon atoms arranged in the form of a convex polyhedral cage. Buckyballs are named after the architect Richard Buckminster Fuller because they look like the geodesic domes that he invented. The first buckyball to be discovered (by accident) was C60, in which 60 carbon atoms are arranged at each of the vertices of a truncated icosahedron. This shape, which looks like a soccer ball, has 32 faces, of which 20 are regular hexagons and 12 are regular pentagons.
Other types of buckyball are known. Common ones have 70, 76, and 84 carbon atoms but all are built up exclusively from hexagonal and pentagonal faces in arrangements that follow Euler's formula. This formula ensures that while the number of hexagon can vary from one type of fullerene to another, every fullerene has exactly 12 pentagons. (In fact, buckyballs with heptagonal faces have been seen, but these faces are concave and are regarded as defects.)


