citric acid
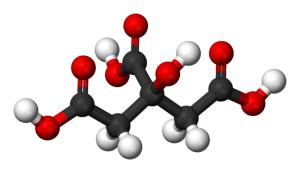
Citric acid molecule. Ball-and-stick model.
Citric acid is a white or colorless, crystalline, sour-tasting hydroxy-carboxylic acid that is present in citrus fruits, such as oranges and lemons, and is an intermediate in the citric acid cycle (Krebs cycle) in plant and animal cells. Its chemical formula is HOOCCH2C(OH)– (COOH)CH2COOH, or C6H8O7. Relative molecular mass 192.1, relative density 1.54, melting point 153°C.
Citric acid is made commercially by fermentation of crude sugar with the fungus Aspergillus niger and is used for flavoring, in effervescent salts, and as a mordant (color-fixer) in dyeing.
Citrate
A citrate is a compound of citric acid and a base. In the body, citrate is involved in blood coagulation (clotting) and the synthesis of fatty acids.


