THE HEALTH REVOLUTION: Surgery and Medicine in the Twenty-first Century 4. New Parts for Old
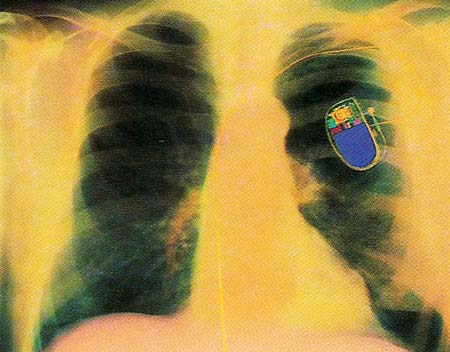
Figure 1. A colored X-ray of a patient's chest, showing a fitted pacemaker. This electronic battery-run device is seen above the rib cage, with a yellow lead that connects it to the heart.
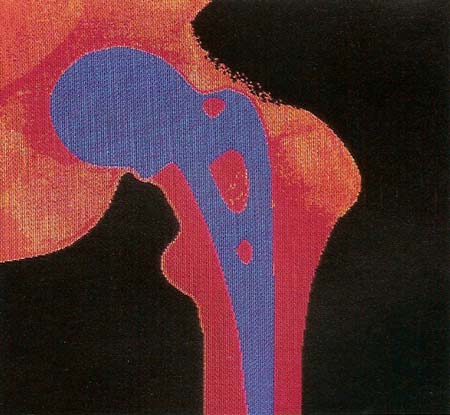
Figure 2. False-color X-ray of a patient's pelvis, showing an artificial hip joint.
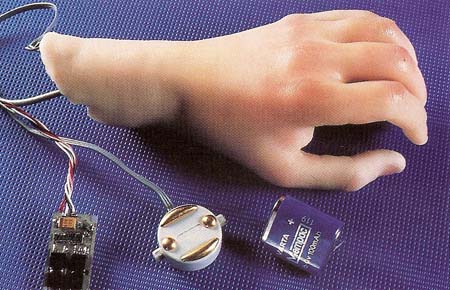
Figure 3. An artificial hand together with the electronic components and wiring that enable it to be connected to the patient's arm muscles.
When a part of the human body stops working through accident or disease, there is a good chance that modern surgery can replace it with either a natural or artificial substitute. The first transplant of an organ from one person to another took place on June 17, 1950, at a hospital in Chicago. A kidney from a woman who had just died was given to a 49-year-old woman named Ruth Tucker. Ten years later, in Cape Town, South Africa, Dr. Christiaan Barnard carried out the world's first heart transplant.
Since then, doctors have learned how to successfully transplant other organs, such as the liver, lungs, and pancreas. Meanwhile, researchers have developed a wide range of implants and prostheses – artificial body parts – to replace everything from hip joints to hands and feet.
Race Against Time
The biggest problem facing surgeons in transplanting an organ from one person (the donor) to another (the recipient) is tissue rejection. A transplant is rejected when the recipient's immune system attacks the cells of the transplant as it would any foreign invader, such as a virus or other disease-causing germ. Components of the blood known as white blood cells surround the cells of the foreign tissues and destroy them. If the body's defense system successfully attacks, or rejects, the cells of a transplanted organ, the organ does.
To reduce the chance that this will happen, organs from potential donors are tested to see if their chemical properties are similar to those of the recipient. This step, known as tissue matching, helps to ensure that the recipient's defense system does not reject the transplanted organ.
Information about patients waiting for organ transplants, including their tissue type, is stored in a computer data bank. Then, when a donor becomes available – often following a fatal accident – the tissue type of the donor is determined and quickly matched against those of a number of possible recipients. If a match is found, the donor's organ is transported to the recipient's hospital. Donated organs must be healthy and preferably young, and they must be delivered to the transplant surgeon as rapidly as possible. After being removed from the donor, the organ is quickly pumped full of fluid to prevent blood clots from forming. Then it is packed in ice and put in a cold storage container at 40°F (5°C).
Speed is essential in transporting the organ to the hospital where the transplant will be carried out. An ice-packed heart becomes useless after 3 to 5 hours, a liver after 10 hours, and a kidney after 24 to 48 hours. Researchers are working to develop special chemicals and new forms of storage that will extend the life of donated organs to many days or even weeks.
Mending Broken Hearts
Far more people new need organs than there are donors available. For this reason, one of the long-range goals of medical technology is to develop complete artificial substitutes for various organs, especially the heart.
During the 1980s, there were 90 people around the world who were fitted with a type of artificial heart called the Jarvik-7, which worked by means of compressed air. One American man, who received his new heart in 1986, survived for 20 months. But the suffering he went through after his operation received a lot of publicity. Doctors and other people debated about whether any more Jarvik hearts should be implanted in patients until more advanced technology became available. Then, at the end of 1989, artificial heart surgery was banned in the United States.
Since that time, researchers have concentrated mainly on improving devices that will assist a damaged heart or take the place of a small section of the heart or blood vessel that is no longer able to do its job properly. For example, diseased arteries – large blood vessels that carry blood away from the heart – can be replaced by new ones made of polyurethane. Damaged heart valves that regulate the flow of blood into and out of the heart and between the heart's chambers can be replaced by valves made of plastic or metal alloy. Meanwhile, scientists continue research to reach the long-range goal of developing total artificial substitutes for such organs as the heart. The artificial organs, however, must be perfected so that they are acceptable to surgeons, patients, and society.
Setting the Pace
The most common implant used to help the heart is the artificial pacemaker (see Figure 1). Normally the heart has its own pacemaker, which produces an electrical impulse along special conducting fibers. When this wave of electrical stimulation spreads across the heart tissue, it causes the muscles of the heart to contract in sequence, pumping blood around the body.
Some diseases interfere with the functions of the heart's natural pacemaker. In such a case, the heart is unable to contract properly. One treatment of this condition involves the implanting of an artificial pacemaker. The artificial pacemaker takes over the job of the defective natural one and, thus, restores normal heart function. The artificial pacemaker is a small electrical unit that is installed under the skin of a patient's chest. Wires from the pacemaker are connected to the heart itself. The wires carry electrical impulses that stimulate the heart so that it beats normally.
Great progress has been made since the first artificial pacemaker was constructed in 1958. The batteries used to power the earliest models had to be replaced every two years or so. In contrast, in 1988 a 47-year-old man was fitted with a nuclear pacemaker containing a tiny pellet of the radioactive material plutonium. The pellet held enough energy to run the pacemaker for at least 20 years. Microprocessors of ever-increasing power are also being attached to pacemakers so that the stimulating signals sent to the heart can be finely adjusted from one beat to the next, depending on the heart's condition. In the future, artificial pacemakers will be able to perform almost as well as their healthy, natural counterparts, while the fuel sources that supply them will last a lifetime.
Artificial Joints
Joints may wear out or become diseased as people grow older, and limbs may become paralyzed through illness or accident. In such cases, modern surgery can often come to the rescue with an impressive array of high-technology replacement parts.
One of the most common operations today is the total replacement of a hip joint (see Figure 2). Every year over 100,000 people in the United States and a similar number in Europe are given new hip joints made of steel and a special hard-wearing plastic. The operation involves gouging out the socket of the joint and replacing it with a plastic cup. At the same time, the head of the femur – the long bone in the upper leg whose ball fits ito the hip socket – is removed and replaced by a metal head. Extending from this metal head is a long stem, which is cemented into a hole drilled into the rest of the femur.
Developing a successful artificial joint such as a hip replacement is much more difficult than it seems. A natural joint can perform well for 70 to 80 years and has its own lubrication system that makes the areas of contact slippier than ice on ice. To provide an adequate substitute, surgeons and engineers must try out all sorts of designs and combinations of materials until they find an arrangement that will do the job. In the future their efforts will be helped by increasingly powerful computers that can accurately model the forces joints are subjected to and also by the creation of new materials that will extend the lifetime of replacement parts.
| Walking With the Computer's Help |
|---|
| Some people lose the use of their legs because of spinal injuries. However, many of these people can now be helped to stand and walk again by using a new computer-aided technique. Electrodes are fastened to a patient's legs, and braces are strapped on to support each knee joint. Wires run from the electrodes to a computer-controlled device that can deliver short electric pulses. Every time a pulse is sent out along a wire, it stimulates the movement of one particular muscle in one of the patient's legs. The computer is programmed with the sequence in which pulses must be transmitted to cause first one leg to take a step and then the other. Progress in learning to walk using this system is often difficult and slow, but it offers real hope to patients who might otherwise be confined to a wheelchair for the rest of their lives. |
Restoring Movement
Victims of accidents who suffer damage to their limbs as well as people paralyzed for various reasons can now benefit from some extraordinary developments in the design of artificial arms, legs, hands, and feet (see Figure 3). For example, people who have undergone amputation of an arm above the elbow can be given an artificial arm that works under electronic control. Tiny electrodes are attached to muscles in the upper arm or shoulder. These electrodes conduct signals from the muscles to the artificial arm so that it can be moved up and down. In the same way, signals can be conveyed from the muscles to vary the firmness of grip of an artificial hand so that it can be used, for example, to pick up an egg or a delicate glass without crushing it.
Equipped with the latest kinds of artificial limbs, people are able to walk, run, lift, and carry just as if they had natural arms and legs. They can even be given a sense of feeling and touch in their new limbs.
The most advanced kind of artificial leg uses devices called transducers, which are placed in the sole of the artificial foot. These transducers send electrical signals to electrodes attached to the skin of the wearer's stump. As many as eight electrodes, each connected to a separate transducer, are attached to a special stump socket that fits precisely against the person's remaining thighbone and muscles. The healthy muscles and nerves in the stump pick up the transducers' signals and transmit them to the brain as if they were coming from a natural foot. The brain interprets the messages as varying degrees of pressure on different parts of the sole. This gives the wearer of such an artificial leg a limited sense of feeling, which in turn allows the person better control over leg and foot movements.
Future developments in the field of artificial limbs will allow users more sensitive feelings in their artificial hands and feet. They will be able to detect changes in texture and temperature and may eventually find it hard to tell the difference between the living parts of their bodies and those that are artificial.

