UP, UP, AND AWAY: The Science of Flight - 1. Amazing Air

Figure 1. Boeing 747 taking off.
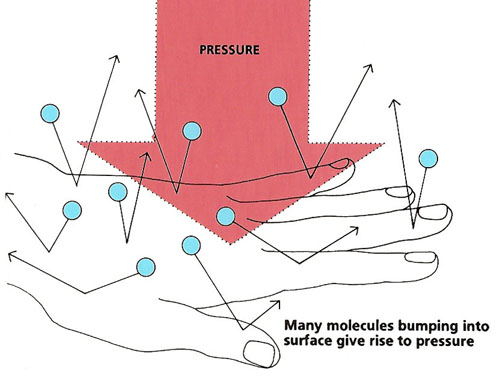
Figure 2. Many molecules bumping into a surface, such as your hand, create pressure.

Figure 3. This drawing shows how Bernoulli's principle results in an upward-pressing force.
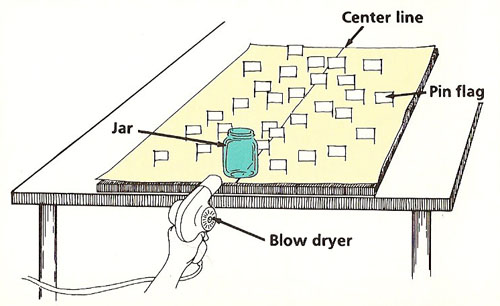
Figure 4. Air flow experiment.

Figure 5. Coanda effect.
A Boeing 747 "jumbo" jet taxis onto the runway, carrying over 350 passengers and crew. Its main deck, 180 feet in length, is longer than the first flight of the Wright brothers in 1908. Its weight is about 400 tons, including enough fuel to drive a car nonstop for about three years. Yet, within seconds, this monster of metal, fuel, and cargo is in the air (see Figure 1). Soon it is flying higher than Mount Everest at a speed of almost 600 miles per hour. How is this possible? How could something so big and heavy fly so well?
An albatross wheels lazily over the ocean, its five-foot-long wings hardly ever beating. Forty thousand above it, a Concorde airliner tears through the sky at twice the speed of sound. In just two and a half hours it whisks its passengers from London to New York. All around the world, there are things flying, hovering, gliding drifting, or floating in the air. Some are living creatures; others are human-made machines. How does each of them manage to stay up? How does flight work?
The answer has to do with air and the way air moves around objects.
Some Pressing News
Air is everywhere around us. It is invisible and, most of the time, unnoticeable. But the power of moving air can make itself frighteningly clear. During a tornado or hurricane, trees are ripped up, houses are wrecked, and cars tossed around as if they were toys.
Air seems weightless. But the air filling your bedroom weighs roughly the same as you do. In fact, all the air on earth weighs about 11 quintillion pounds!
Like most matter, air is made up of tiny particles called MOLECULES. In solids or liquids, the molecules are packed tightly together. But in gases such as air the molecules are far apart and move very rapidly. The average speed of air molecules in a warm room is about 1,130 miles per second.
When an air molecule crashes into something, such as your hand, it gives it a tiny push before bouncing off again. The effect of just one molecule is far too small to notice. But billions and billions of air molecules bump into your hand every second (see Figure 2). That gives rise to quite a strong pressing force, or PRESSURE. Why, then, do you not feel it? The answer is that more or less the same number of molecules are colliding with the other side of your hand. So, the pressure on either side is balanced out.

Air on the Move
You will need:
What to do:
Hold one of the sheets of paper close to your bottom lip and blow hard across the upper surface. What happens?
Place the two books about 4 inches apart on a table. Lay a sheet of paper over the books. Blow hard through the gap between the books. Notice how the paper moves.
Hold the two sheets of paper upright, a few inches apart in front of your face. Again, blow hard and watch what happens.
Try to explain your observations by thinking about the air pressure on either side of the paper in each of the experiments. What did you do to the air on each side when you blew on it? What effect do you think this had on the pressure on that side? |
More Speed, Less Pressure
In 1738, the Swiss mathematician Daniel Bernoulli made a surprising discovery. It has become known as BERNOULLI'S PRINCIPLE.
Bernoulli found that as the speed of a gas or liquid increases, its pressure drops. This means that air rushing over a surface, for example, pushes against the surface less than if the air were still.
If air moves everywhere around an object at the same speed, then the pressure on all sides will drop by the same amount. In this case, the pushing force from every direction remains balanced. But what happens if the air only moves over the top of an object and is still underneath? From Bernouilli's principle it follows that the pressure on top will be less than that underneath. Because of this, there will be an upward-pressing force (see Figure 3).

Mapping Air Flows
You will need:
What to do:
Cover the board with the sheet of paper. Draw a line down the middle of the paper at right angles to the edge nearest you.
Cut a 1/2-inch wide, 2-inch long strip of paper and fold it around one end of the pins. Glue the ends of the paper together so that the result is a small flag, with the pin as the flagpole. The paper should be able to spin easily around the pin when you blow on it. Make similar flags using the other pins.
Place the glass jar at the near edge of the board on the middle of the line. Stick the pins in at various points behind and to the side of the jar. Some should be close behind the jar, others farther away. Turn all the flags so that they are pointing inward at right angles to the center line.
Point the blow dryer at the near side of the jar, about 6 inches away, and directly along the center line. Switch it on to the lowest setting for several seconds (see Figure 4). Then turn it off. Look at the new position of the flags.
Under each flag draw a short line to show the direction in which it now points. Then remove the flag. Observe the pattern of lines. What does this tell you about the way the air flowed around the jar?
Taking it further:
Repeat the experiment with the pins in new positions. For example, you could stick the pins at the ends (or beginnings) of the short lines. By doing this a number of times, you could map out complete lines of flow. Make sure that you hold the blow dryer in exactly the same place in each time.
Do the whole experiment again with different shaped obstacles – some with straight sides, others with smoothly curved sides. What happens if you make the surface of he jar less smooth by wrapping it with cloth? What happens if you increase the speed of the air flow? |
Sticky Streams
Sometimes when you try to pour water from a jug the water runs down the side of the jug. In fact, all liquids and gases behave this way (see Figure 5). That is, when flowing past a smooth surface, they tend to stay close to the surface. The effect was first described by a Romanian engineer, Henri Coanda, in 1926, and is named after him.
Because of the COANDA EFFECT, moving air will try to follow the lines of an object, even if it has to change direction to do so.

