BETWEEN FIRE AND ICE: The Science of Heat - 1. Hot Subject

Figure 1. Everything contains heat: the icy slopes of this volcano as well as its hot inside.
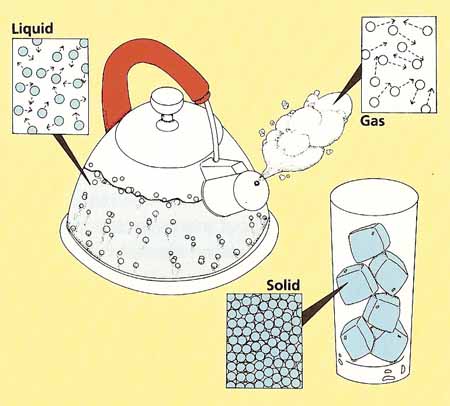
Figure 2. Molecules in a solid, a liquid, and a gas.
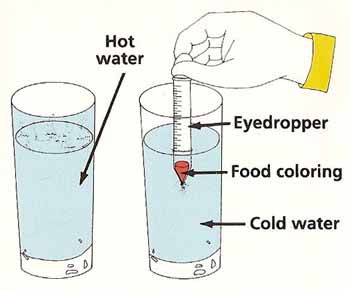
Figure 3. Liquid mix-up experiment.
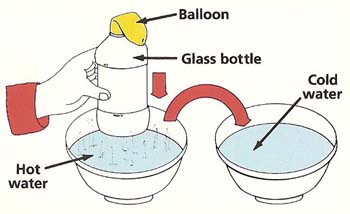
Figure 4. Balloon experiment.
Heat is everywhere: in volcanoes, in icebergs, in your body – in every object in the universe (see Figure 1). All matter contains heat.
Like light and sound, heat is a form of ENERGY. This means that it can be used to do work and cause movement. It can also be transferred from one object to another. For example, some of the heat tat starts out in the center of the Sun eventually reaches the Earth and provides us with the warmth we need to live.
Moving Molecules
Almost all the substances round you are made up of MOLECULES. These are very tiny particles that never stop moving.
Whenever you breathe in deeply, your lungs fill with about 5 septillion (5 and 24 zeros following it) molecules of oxygen and about 18 septillion molecules of nitrogen. These molecules are racing around at an average speed of about 1,500 feet per second, the speed of a rifle bullet.
As air is heated up, the molecules in it move faster. As it is cooled down, the molecules move slower.
In a gas or a liquid, the molecules are able to move around freely. In a solid, the molecules are stuck together. However, they can still vibrate back and forth. As a solid is heated up, the vibrations get bigger and faster.
In any substance, whether it is a solid, a liquid, or a gas, the speed of the molecules is a measure of how hot or cold it is.
Molecules in Collision
In a liquid or gas, the molecules behave like bumper cars (see Figure 2). They keep ramming into one another and bouncing away again. If two different liquids are put together, their molecules immediately start to collide and become mixed up. If one of the liquids is hot, its molecules will smash hard into the molecules of the other liquid and cause them to spread out quickly. On the other hand, if both liquids are cool, their molecules will move more slowly and take longer to mix together.

Liquid Mix-UpYou will need:
What to do:
Fill one of the glasses with cold water and the other with hot water. Suck up a small amount of food coloring into the eyedropper. Put the end of the eyedropper in the middle of the glass of cold water and gently squeeze out a drop of food coloring. Do the same in the glass of hot water (see Figure 3). Compare what happens in the two glasses. Try to explain what you see. |

Hot Air, Cold Air
You will need:
What to do: Take the cap off the bottle and place the balloon over the top. Fill one of the bowls with cold water and the other with hot water. Place the bottle in the hot water (see Figure 4). Does anything happen to the balloon? Now place the bottle in cold water. What happens? Try to explain your observations. |
Heat and Gases
As a gas is heated up, its molecules move faster. They collide harder and more often among themselves. They also collide harder and more often with the walls of their container. If the container is made of a stretchy material, such as rubber, it will expand as the gas inside gets hotter. If the container is stiff, as the gas inside heats up, its molecules will not be able to make the container bigger. Instead the gas molecules will just push harder and harder against the walls.

