BETWEEN FIRE AND ICE: The Science of Heat - 2. Shrink and Swell

Figure 1. This outdoor thermometer shows that it is a hot day.
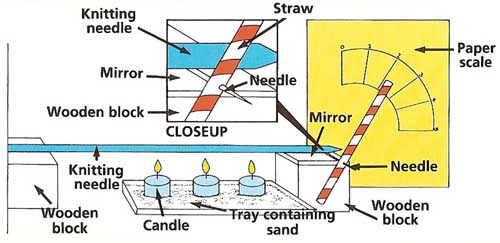
Figure 2. metal expansion experiment.
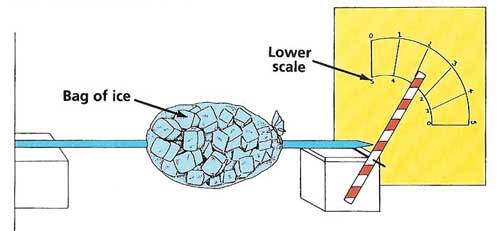
Figure 3. Liquid mix-up experiment.
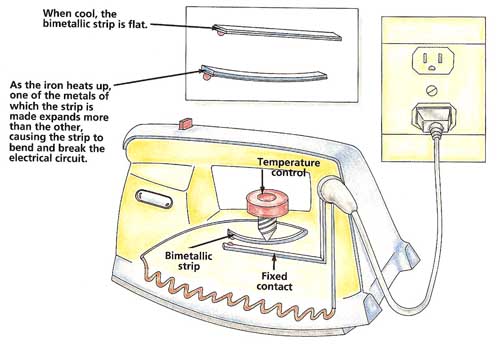
Figure 4. How a clothes iron works.

Figure 5. An oral thermometer.
On a hot summer day, telephone wires sag between the poles that hold them up. Yet, on a cold day, the wires are tightly stretched. The reason is that most substances expand as they get hotter, and contract, or shrink, as they cool down (see Figure 1).
Metals, when heated, expand more than other kinds of solids do. Because of this, engineers have to be especially careful when designing and building large metal structures. For example, narrow gaps must be left between railroad tracks at regular intervals. This prevents the tracks from buckling during hot weather.
 |
|---|
Lengthening RodsYou will need:
What to do: Stick the sewing needle through the straw about a third of the way from one end. Place the mirror on top of one of the wooden blocks. Lay the knitting needle across the two wooden blocks so that one end is touching a wall. Place the needle holding the straw between the mirror and the free end of the knitting needle. Make a paper scale as shown and affix it to a wall of wooden board to show the position of the straw pointer. Arrange the straw so that its long end points straight up at the zero reading. Place the three candles in a line on the baking tray so that they are directly under the knitting needle (see Figure 2).
With the help of an adult, light the candles and start the stopwatch. Note the pointer reading at 1-minute intervals until the reading does not change anymore. Blow out the candles. Plot a graph of pointer readings against time. Can you draw any conclusions from this graph?
Taking it further: Repeat the experiment using the rods made of different metals. Make sure the test is fair by heating each rod for the same amount of time. Which metal expands the most? Plot graphs of pointer readings against time for each metal. Do all the graphs look the same? (see Figure 3)
Allow the metal rods to cool down and return to their original length. At the bottom of the paper scale, mark a new set of readings, as shown, that run in the opposite direction to the top set. Poke a hole in the side of the bag of ice with one of the metal rods. Push the rod all the way through the ice bag and out the other side. Rest the ends of the rod on the wooden blocks as before. Move the straw so that it is pointing at zero on the bottom scale. Start the stopwatch. Note the readings on the bottom scale at 1-minute intervals. What happens to the rod? Draw a graph of your findings. Repeat the experiment using the other rods and compare your results. Does the rod that expanded the most also contract the most?
Although metals expand more than other solids, the amount by which they expand is still very small. For example, iron expands by 0.0001 (one ten-thousandth) of its length for every degree Celsius rise in its temperature. This means that an iron rail 45 feet long would become just over 1½ inches longer if its temperature rose by 86°F (30°C). The gaps between rails on a railroad track are to allow for this.
In our experiment, the increases in length of the rods are much less than an inch. For example, if an iron or steel rod starts out 12 inches (30.4 cm) long and has its temperature raised by 212°F (100°C), it will lengthen by only 0.12 inches (0.3 cm). This is such a small amount that it would be difficult to observe it if a pointer were simply fixed to the end of the rod. By using the needle, mirror, straw, and curved scale as described, however, the amount by which the rod grows is magnified. This also makes it easier to observe differences in the expansion of different metals. Such methods of making small effects more noticeable are often used in experiments and in measuring equipment. |
Making Expansion Work
The fact that different metals expand by different amounts can be put to good use. Inside some appliances are two thin strips of different metals joined together to make what is called a "bimetallic strip." When cold, the bimetallic strip is straight. When heated, however, the metal that expands more bends the strip into a curve. This can be used to make or break an electrical connection.
In a clothes iron, for example, a bimetallic strip bends as the iron heats up (see Figure 4). Eventually, the strip touches another piece of metal. This completes an electrical circuit that opens a switch so that the iron does not overheat. The position of the strip can be altered by a control to set the temperature of the iron. The same means is used to control central heating systems.
Measuring Hotness
 |
Like solids, liquids and gases also expand when heated and contract when cooled. The amount by which a substance expands or contracts depends on how much its temperature changes. Temperature is the degree of how hot or cold something is, measured with a THERMOMETER (see Figure 5).
Most thermometers consist of a narrow, sealed glass tube with a bulb at the end. Inside the bulb is a liquid. In some cases, this is mercury, a silvery liquid metal. The hotter the mercury gets, the more it expands and moves up the tube. Along the tube is a scale. This may show the temperature in degrees Fahrenheit or in degrees Celsius (centigrade). Fahrenheit is used for everyday purposes in the United States. In other parts of the world, though, temperature is normally given in Celsius.
The level the mercury reaches in melting ice is fixed at 0°C or 32°. In boiling water, a thermometer shows 100°C or 212°F. In between these two fixed points, the scales are laid out in equally space divisions.
Thermometers containing alcohol are also common. The alcohol, which is normally clear, is colored red so that it shows up more easily. Alcohol is less expensive than mercury and can be used to measure lower temperatures.
 |
|
|---|---|
Taking TemperaturesYou will need:
What to do:
Set up the outdoor thermometer in a shaded place outside your house or school. Record the temperature every half hour from early morning to late afternoon or early evening. Plot a graph of the results. How did the temperature vary during the day? What were the lowest and highest temperatures? Repeat this experiment on other days. Do the daily changes in temperature always follow the same pattern?
Use the indoor thermometer to measure the temperature inside your house or classroom. How does the reading change if you put the thermometer near a window, especially when the Sun is shining through? Leave the thermometer in the refrigerator for 15 minutes and record the temperature. Do the same in the freezer.
Ask an adult to help you use the oral thermometer (see Figure 5). This must be shaken first to make sure that all the mercury is returned to the bulb. Place the thermometer carefully under your tongue and leave it there for 3 minutes. Check the reading. Find out what the temperature of a healthy person should be.
Taking it further: With a teacher's help, carry out a survey of the temperatures of people in your class. After each person has had his or her temperature taken, the thermometer must be sterilized. That is, it must be properly cleaned to remove any germs. Your teacher will show you how to do this. Record everyone's temperature to the nearest tenth of a degree. You will probably find that there is some variation in the readings. Count how many people are in each tenth-of-a-degree range from 98° to 99°F (36.3° to 36.9°C). Plot a bar chart to show your results. From this, what would you conclude is a normal temperature range for human beings? |
Heat and Temperature
There is an important difference between how hot something is and how much heat it contains. You will already have discovered this if you have bitten into an apple pie from a fast-food restaurant. Although the pastry on the outside and the apple on the inside are at nearly the same temperature, the pastry is easy to eat while the apple can burn your mouth. The reason is that the apple contains much more heat.
 |
|---|
The Speed of CoolingYou will need:
What to do: Place one thermometer in the pan and ask an adult to heat the water in the pan to a temperature of about 160°F (70°C). Ask the adult to put the pan on a cool surface. Read the temperature and start the stopwatch. Take the temperature every minute for half an hour. Using the second thermometer take the temperature of the room. Plot a graph of how much the water cooled each minute (the rate of cooling) against the difference between the water temperature and the temperature of the room. What shape is the graph? What can you deduce from this?
Taking it further: This experiment can be extended to other substances. For example, what happens to the shape of the cooling curve if you use hot coffee, syrup, or hot cereal? In the section 'Heat and Temperature', the point is made that some things contain more heat than others, even though they are at the same temperature. By comparing the way different liquids, such as water and hot cereal, coo, can you say which contain the most heat? Try to explain the reason for this. |
Newton's Law
Several hundred years ago, Sir Isaac Newton made an important discovery about how fast things cool down. He found that the rate of cooling depends on the temperature difference between an object and its surroundings. In other words, the hotter something is, compared with its surroundings, the faster it will cool down. This is known as Newton's Law of Cooling.
 |


