Balmer series

The Balmer series is a series of emission lines or absorption lines in the visible part of the hydrogen spectrum that is due to transitions between the second (or first excited) state and higher energy states of the hydrogen atom. It is are named after their discoverer, the Swiss physicist Johann Balmer (1825–1898).
The transition from the third level to the second level yields the red H-alpha emission line at 6563 Å; H-beta is in the green part of the spectrum at 4861 Å, H-gamma is in the violet (as are higher members of the series) at 4342 Å, and H-delta at 4101 Å. The Balmer jump is the relatively abrupt decrease in a continuous spectrum at about 3650 Å caused by hydrogen absorption lines in the Balmer series crowding to their series limit.
A formula developed by Balmer in 1884 relates the frequencies of these lines to one another. In terms of wavelength λ, the formula is:
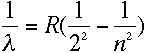
where R is Rydberg's constant, whose value is approximately 1.097 × 107 m-1, and n is 3, 4, 5, 6, or 7 for the five lines studied by Balmer.


