chelate
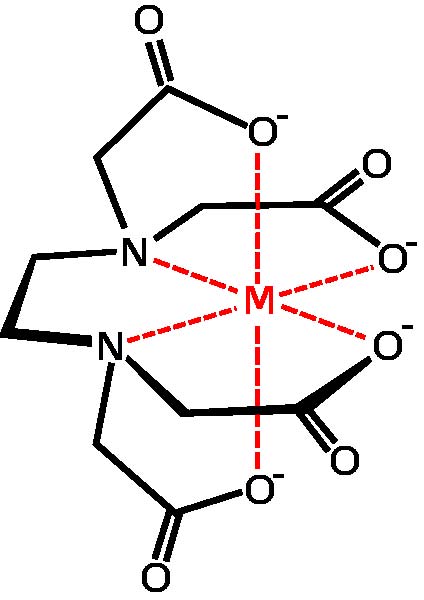
A chelate is a chemical complex formed from a polydentate ligand and a metal ion, thus making a ring. Chelates are more stable than the corresponding unidentate complexes, and are used to sequester metal ions (see hard water), as well as in chemical analysis and for separating metals. Some biochemical substances, including chlorophyll and hemoglobin, are chelates (see porphyrin).
A chelating agent is a substance that helps form chelates by binding metal ions. Some chelating agents, including desferrioxamine and penicillamine are drugs used to treat metal poisoning; the metal is bound to the drug and excreted safely. Chelating agents often form the active centers of enzymes.


