heat transfer
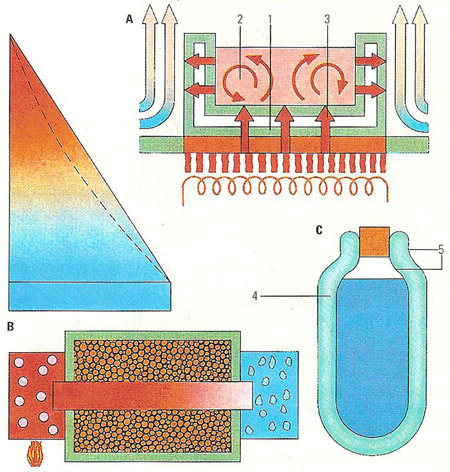
The three ways in which heat moves all take place when a pan is heated (A) – conduction through the metal walls of the pan (1), convection by fluid motion (2), and radiation from the heat source to the pan (3). In theory an insulated good conductor with ice at one end and boiling water at the other varies in temperature linearly with distance along the bar (B), as in the straight-line graph. With poor insulation a curve like the dotted line results. A vacuum flask (C) has a vacuum (4) to prevent conduction and convection and silvered walls (5) to minimize heat loss by radiation.
Heat transfer, also called heat exchange, is a flow of heat energy from one object to another. This flow of energy occurs at all times when two or bodies at different temperatures are in thermal contact. Three methods of heat transfer can be distinguished: conduction, convection, and radiation. In conduction, heat is transferred from molecule to molecule within a body, as in an iron rod stuck in a fire; in convection, heat is transferred by circulation of a fluid, as in boiling; in radiation, heat is transferred in the form of electromagnetic waves, as in sunlight.
Heat exchangers are essential in many industrial processes where heat may be extracted from one source for use elsewhere without the two sources combining. The most simple heat exchangers use conduction to achieve this. For example, a hot fluid is run through tunes running parallel to one another (and with a high surface area). This arrangement of tubes is immersed or built around a container, into, and through, which another, cold fluid flows. By conduction the heat is transferred from the hot fluid to the cold.


