catalysis
Catalysis is the changing of the rate of a chemical reaction by the addition of a small amount of a substance which is unchanged at the end of the reaction. Such a substance is called a catalyst, though this term is usually reserved for substances which speed up reactions; additives which slow down reactions are called inhibitors.
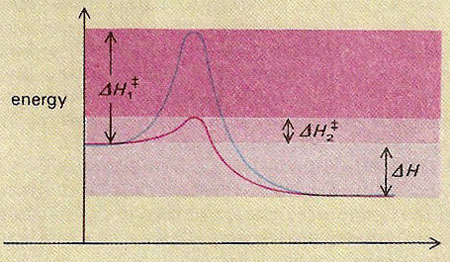
In general, chemical reactions are possible if the reaction products have lower energy than the reactants. But the reaction only proceeds if sufficient energy is available to exceed the activation energy (ΔH1) for the reaction. Many catalysts work by providing a reaction pathway which has a lower activation energy (ΔH2) than the uncatalyzed reaction. The overall heat of reaction (ΔH) remains the same.
Autocatalysis
Autocatalysis is a form of catalysis in which one of the products of a reaction serves as a catalyst for the reaction. Before the emergence of protein-based enzymes, which are the principle biological catalysts today, it is believed that a substance, such as RNA, may have served as both a replicator and an autocatalyzer in the first terrestrial life-forms.


