lignin
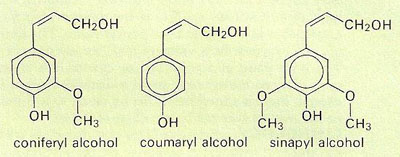
Lignin is a complex polymer formed by condensation of precursors such as coniferyl, coumaryl, and sinapyl alcohols.
Lignin is a complex aromatic polymer that forms 25–30% of the wood of trees and is also found in all vascular plants, mostly between the cells, but also within the cells, and in the cell walls. It makes vegetables firm and crunchy. It functions to regulate the transport of liquid in the living plant (partly by reinforcing cell walls and keeping them from collapsing, partly by regulating the flow of liquid), and enables trees to grow taller and compete for sunshine. Lignin is deposited in the secondary cell walls of schlerenchyma, xylem vessels, and tracheids. After cellulose, lignin is one of the most common organic substances on Earth. In making paper the lignin must be separated from the cellulose.
Lignin typically stains red in prepared microscope slides. In foods, lignin is considered dietary fibre. There is evidence that lignin binds to carcinogens and other harmful substances and makes them safe.
Lignin chemistry
Lignin is not one compound but many. All are complex, amorphous, three-dimensional polymers that have in common a phenylpropane structure, that is, a benzene ring with a tail of three carbons. In their natural unprocessed form, they are so complex that none of them has ever been completely described, and they have molecular weights that my reach 15,000 or more.
Lignins as they occur in nature (protolignins) have been grouped into several types, characteristic of hardwoods, softwoods, and grasses. Within each type there is a lot of variation: lignins differ from species to species, and from one tissue to the next in the same plant – even within different parts of the same cell. The process of removing them from the plant changes their form and chemical makeup to a greater or lesser extent, which makes then hard to study and way account for the large and growing number of analytical techniques in use.


