astatine
Astatine (At) is a semimetallic (see metalloid), highly unstable radioactive element (atomic numer 85) that resembles iodine in solution and accumulates in the thyroid gland. Astatine is a member of the halogen family of elements. It occurs naturally in minute quantities, and can be prepared by bombarding bismuth with alpha particles.
Astatine has 33 known isotopes, the longest lived of which is At-210, with a half-life of 8.3 hours. At-211 (half-life 7.2 hours) is used in medicine as a radioactive tracer.
Astatine was discovered by Dale R. Carson and colleagues in 1940. Its name comes from the Greek astatos for "unstable".
| atomic number | 85 |
| relative atomic mass | 210 |
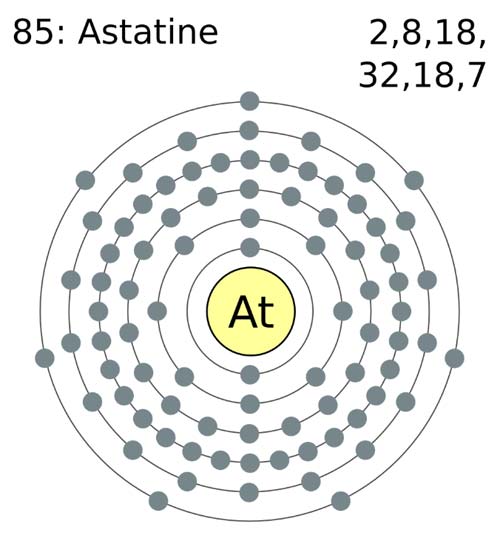
| electron configuration | [Xe]4f141s25d10626p6 |
| first ionization energy | 890±40 kJ/mol |
| melting point | 302°C (576°F) |
| boiling point | 337°C? (639°F?) |


