cellulose
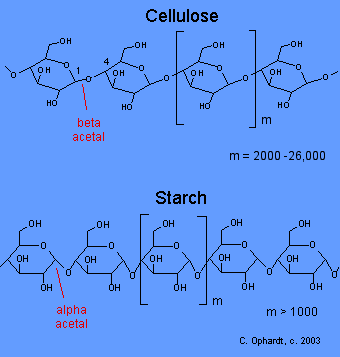
The molecules of cellulose and starch are very similar.
Cellulose is a polysaccharide that is the main constituent of the cell wall in all green plants and most bacteria. Cellulose is the most abundant compound on Earth that is manufactured by living things.
Cellulose and starch are both made from glucose monomers (building blocks) but there is a crucial difference between them. The monomer in cellulose is beta-D-glucose and the chemical link between these monomers is called a beta acetal linkage. As a result of the bond angles in the beta acetal linkage, cellulose is mostly a linear chain. By contrast, the bond angles of the alpha acetal linkage found in starch gives rise to a spiral structure like a coiled spring. This difference in acetal linkages has a profound effect on digestibility.
Humans are unable to digest cellulose because the appropriate enzymes to breakdown the beta acetal linkages are lacking in our bodies. Indigestible cellulose is the fiber which aids in the smooth working of the intestinal tract. Animals such as cows, horses, sheep, termites, and soil bacteria, however, possess the necessary enzymes to digest cellulose.
When processed cellulose is used in many different ways: wood for building; paper products; cotton, linen, and rayon for clothes. It is also in manufacturing cellophane, celluloid, and the cellulose plastics, notably nitrocellulose (used in explosives), cellulose acetate for toys, boxes, and films, and cellulose acetate butyrate for typewriter keys.
Cellulase
Cellulase is any of a group of enzymes, produced mainly by fungi, bacteria, and protozoans, that is able to break cellulose into smaller molecules, primarily sugars. There are several different kinds of cellulase, which differ in their structure and the way they act, but basically all bring about the hydrolysis of cellulose. In the best-known example of cellulase activity, cellulose is broken down into β-glucose by cellulase produced by symbiotic bacteria in the stomachs of herbivores. The basic mechanism involved is the cleaving of β-glycosidic links the connect the sugar units of cellulase.
Cellulose plastic
Cellulose plastic is a thermoplastic derivative of cellulose. Cellulose plastics include nitrocellulose (cellulose nitrate), used in explosives and propellants; various cellulose acetates for making textile fibers and packaging films; and ethyl cellulose, used in the manufacture of shock-resistant materials.


