coacervate
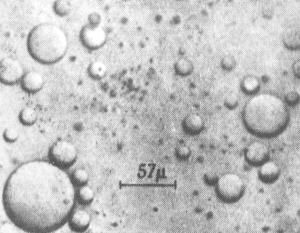
Coacervate droplets formed by interaction between gelatin and gum arabic. A. I. Oparin. Note: This photo appears on p. 103 of The Origin of Life by Cyril Ponnamperuma (E. P. Dutton & Co., 1972).
A coacervate is a spherical aggregation of lipid molecules making up a colloidal inclusion which is held together by hydrophobic forces. Coacervates measure 1 to 100 micrometers across, possess osmotic properties, and form spontaneously from certain weak organic solutions. Their name derives from the Latin coacervare, meaning to assemble together or cluster. It was suggested by Oparin that coacervates may have played a significant role in the evolution of cells.
In water, organic chemicals do not necessarily remain uniformly dispersed, but may separate out into layers or droplets. If the droplets which form contain a colloid rich in organic compounds and are surrounded by a tight skin of water molecules then they are known as coacervates. These structures were first investigated by the Dutch chemist H. G. Bungenburg de Jong in 1932 (Bungenberg de Jong, H. G., Protoplasma 15: 110, 1932.). A wide variety of solutions can give rise to them; for example, coacervates form spontaneously when a protein, such as gelatin, reacts with gum arabic.
Coacervates are interesting in that not only do they provide a locally segregated environment but also that their boundaries allow the selective adsorption of simple organic molecules from the surrounding medium. In Oparin's view this amounts to an elementary form of metabolism. J. D. Bernal commented that they are "the nearest we can come to cells without introducing any biological – or, at any rate, any living biological – substance." However, the lack of any mechanism by which coacervates can reproduce leaves them far short of being actual living systems.


