electroplating
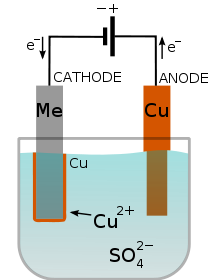
Electroplating of a metal (Me) with copper in a copper sulfate bath.
Electroplating is the process of depositing a thin metal coating on base-metal objects, to improve their appearance or corrosion resistance. The object is made the cathode of a cell containing a salt of the metal to be deposited, which is made the anode; on electrolysis the metal dissolves from the anode and deposits on the object. Chromium, nickel, copper, silver, and gold are commonly used. In electropolishing – the reverse process – the object is made the anode; preferential solution of irregularities yields a high polish.


