silver

Figure 1. Native silver. Credit: USGS / US House of Representatives.
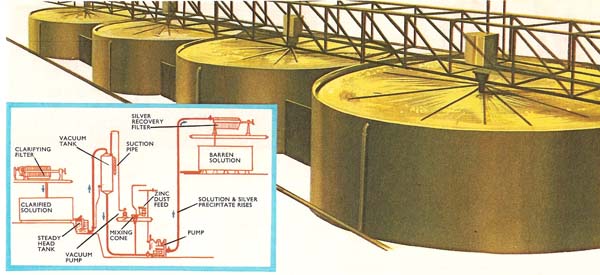
Figure 2. The cyanide process is used for the extraction of both silver and gold. (Above): Large tanks in which the cyanide solution reacts with silver ores. (Left): Flow diagram for the whole process.

Figure 3. In the Parkes process for desilvering lead, zinc (in which silver is much more soluble) is added to the molten lead-silver alloy. After thorough mixing, the zinc-silver layer floats to the top and is skimmed off.

Figure 4. Sheffield plated soup tureen (c. 1815).
Silver (Ag) is a lustrous, white, ductile, malleable, metallic element. It is also a noble metal and a transition element, occurring in group IB of the periodic table. It has the highest thermal and electrical conductivity of any metal.
Silver occurs both uncombined, sa native silver (Figure 1), and in ores such as galena and argentite. (Argentite, also called silver glance, is a soft, dark gray sulfide ore of silver (Ag2S), related to chalcocite. It occurs in veins, often together with native silver, in parts of Norway, Germany, the Czech Republic, South America, Mexico, and Nevada).
Silver is highly valued for jewelry, tableware, and other ornamental uses, and is widely used in coinage, photography, dental amalgam and soldering alloys, and electrical circuits. Although silver is the best conductor of heat and electricity, copper is generally used in its place because it is cheaper. However, silver is sometimes used in chemical plants due to its good resistance to corrosion, comparatively high melting point, and good thermal conductivity. Thus reaction vessels and reboilers in distillation are sometimes lined with silver, and the tubes of heat exchangers may be made of silver. It is also used in various types of electrical apparatus, including contact breakers.
Silver is concentrated by various processes including cupellation and extraction with cyanide, and is refined by electrolysis (see below for details).
| atomic number | 47 |
| relative atomic mass | 107.87 |
| electron configuration | 1s22s22p63p23p63d10 4s24p6 5s14d10 |
| atomic radius | 144 pm |
| relative density | 10.50 |
| melting point | 960.8°C (1,761°F) |
| boiling point | 2,212°C (4,014°F) |
History
Silver sometimes occurs in native form (found as an element in its natural state) and so, along with gold and copper, was one of the first metals knownto humans. For thousands of years it has been valued for its beauty, and silver ornaments and jewelry dating from 4,000 BC have been found in Egyptian tombs. By about 800 BC it was in use as currency over the whole of the Middle East; silver coins were being minted in Greece about 700 BC.
Although silver occurs free in nature, it is much more often found as silver sulfide, mixed with lead sulfide, in the ore galena. As early as 2,500 BC galena was mined and smelted; the silver was separated from the lead by cupellation. In this process the lead is oxidized in a furnace and removed as a scum from the surface of the molten silver which remains behind. The galena which was mined in Greece about 600 BC probably contained about 60 ounces of silver per ton of ore.
Silver was mined in Europe, mainly in Bavaria and Spain, in medieval times. The discovery of the New World, and particularly Mexico and Peru, led to an enormous increase in the world supply of silver during the 16th and 17th centuries. Mexico and Peru continue to be major suppliers.
Extraction
Silver is obtained principally as a by-product when lead, zinc, and copper ores are refined. However, it is also obtained from ores mined for their silver content, the commonest of which is argentite, a sulfide of silver.
Of the many processes which have been developed over the years, the cyanide process is normally used now in extracting silver from argentite (see Figure 2). After the ore has been crushed, it is ground to a fine powder in a ball mill which also contains weak sodium cyanide solution. The silver sulfide and also any free silver present reacts with the sodium cyanide to yield sodium argentocyanide, Na[Ag(CN)2], which is soluble in water. The suspension is agitated with compressed air for several days. This serves to oxidize the sodium sulfide formed by the reaction between silver sulfide and sodium cyanide. The solution of sodium argentocyanide is then filtered and the dissolved air removed by vacuum. The silver is finally displaced from solution by adding a suspension of finely powdered zinc dust.
The precipitated silver is filtered off and dried. This product contains at least 75% silver, but will always contain impurities, particularly the excess of zinc dust. Further purification is, therefore, necessary.
As about three-quarters of the world's output of lead is obtained from ores which also contain silver, it follows that the recovery of silver is a profitable stage in refining lead. One way of doing this is by the Parkes process (Figure 3). After removing other impurities, namely copper, antimony, arsenic, and tin, from the molten lead-silver alloy, zinc is added to the bath. Silver is much more soluble in zinc than in lead, so that after mixing the contents of the bath most of the silver migrates to the layer of molten zinc which floats on top of the lead. When the temperature of the bath is allowed to fall, the zinc-silver alloy is skimmed off as it solidifies on the surface. The zinc can then be distilled off leaving behind the silver which has a higher melting point.
Physical properties
Silver is harder than gold but softer than copper. Apart from gold, it is the most malleable (bendable) and ductile (it can be stretchable) of all metals. One gram of silver can be drawn out into a thin wire 1½ kilometers long, and it can be beaten out into sheets only 0.00025 millimeter thick. It is an excellent conductor of heat – better even than copper – and is also a slightly better conductor of electricity than copper.
Chemical properties
Silver, with few exceptions, is monovalent in its compounds. It is very resistant to attack by oxygen and tarnishes in air only if sulfur compounds are present, when a thin film of sulfide forms on its surface. The film which appears on silver egg spoons is caused by the action between organic sulfur compounds and the silver. It is attacked by the halogens chlorine, bromine, and iodine, but is resistant to attack by most alkalis and acids, except nitric acid and hot concentrated sulfuric acid.
Silver chloride, bromide, and iodide (see below) are all sensitive to light, and for this reason are used in the manufacture of photographic films and papers. It forms alloys, which are used in jewelry and coinage, with copper and gold.
Compounds of silver
Silver halides (AgX) are crystalline salts used in photography. The chloride is white (and occurs naturally as the mineral horn silver), the bromide pale yellow, and the iodide yellow. On exposure to light, a crystal of of silver halide becomes activated, and is preferentially reduced to silver by a mild reducing agent (the developer).
Silver nitrate (AgNO3) is a transparent crystalline solid, the most soluble of the silver salts. It is used in chemical analysis, silver-plating, and dyes, and as an antiseptic and astringent, especially for removing warts. Application discolors the skin bluish-black. Specific gravity 4.3; melting point 414°F.
Sheffield plate
Sheffield plate (Figure 4) is any article made from silver plated on copper by a method of fusion involving heat and pressure, discovered about 1743 by a Sheffield culer, Thomas Boulsover. This method was widely used before the advent of electroplating.


