excitation
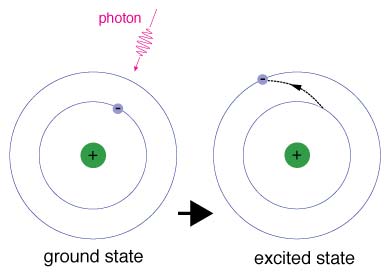
Excitation is the lifting of electrons from lower energy levels in atoms to higher energy levels by the injection of energy. This energy can come from two main sources. In radiative excitation, a photon is absorbed whose energy is equal to the difference between the energy levels. In collisional excitation, the energy needed for a particular electron jump comes from the impact between the atom and another particle. In both cases, when electrons fall back to lower energy levels they give off photons at specific wavelengths.
The excitation temperature of a gas or plasma is the temperature determined by the proportions of atoms or ions in the ground state and in excited states.


