xylene
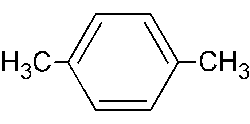
Paraxylene.
Xylene, C6H4(CH3)2, is colorless, sweet-smelling, flammable liquid of the aromatic group of hydrocarbons made by the catalytic reforming of certain naphthenic petroleum fractions. Xylene (dimethylbenzeme) is both naturally-occurring and manufactured by man, and is used as high-octane motor and aviation gasoline blending agents, solvents, and chemical intermediates. It is also used for increasing the transparency of tissues prepared for microscopic examination after dehydration. It occurs as three distinct structural isomers: metaxylene (m-xylene), orthoxylene (o-xylene), and paraxylene (p-xylene).
Before 1940 virtually all aromatic solvents, including xylene, were produced from coal. Thereafter xylene began to be produced from petroleum refining. It also occurs naturally in petroleum and coal tar and is formed during forest fires.
More than 90% of mixed xylene isomers is used for blending into gasoline. The remainder is used in a variety of solvent applications, as well as to produce the individual isomers of xylene. Xylene based solvents are widely used in the paint and printing ink industries, for polyester fibeR, film and fabricated items and perfumes, pesticide formulations, pharmaceuticals and adhesives. It is also used for household products such as aerosol paints and lacquers Xylene is also used as a cleaning agent, and as a thinner for paints and varnishes. Individual isomers of xylene are used in the manufacture of certain plastics.
Commercial or mixed xylene usually contains about 40–65% m-xylene and up to 20% each of o-xylene and p-xylene and ethylbenzene. Xylenes are released into the atmosphere as fugitive emissions from industrial sources, from auto exhaust, and through volatilization from their use as solvents. Acute (short-term) inhalation exposure to mixed xylenes in humans results in irritation of the eyes, nose, and throat, gastrointestinal effects, eye irritation, and neurological effects. Chronic (long-term) inhalation exposure of humans to mixed xylenes results primarily in central nervous system (CNS) effects, such as headache, dizziness, fatigue, tremors, and incoordination; respiratory, cardiovascular, and kidney effects have also been reported.


