azo compound
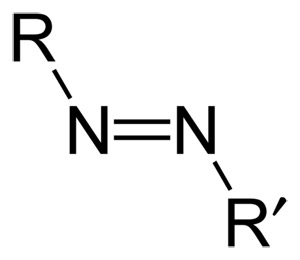
General formula of an azo compound.
An azo compound is any of a class of nitrogenous organic compounds having the general formula R–N=N–R'. The most important azo compounds have aromatic groups for R and R', and are made by coupling diazonium compounds with nucleophiles such as phenols or aromatic amines, or by coupling heterocyclic hydrazines with aromatic amines. They comprise more than half the dyes commercially available.
Many azo dyestuffs are sulfonic acid derivatives and are thus soluble in water. They are used as acid dyes for wool, and their affinity for cotton is increased by the number of auxochromes or azo groups. Alkylation of phenolic groups increases fastness. Azo dyes are used as pigments, in color photography, and as dyestuffs for most materials. They can be used as mordant or pre-metallized dyestuffs. For more on azo dyes, see aniline.


