crystallization
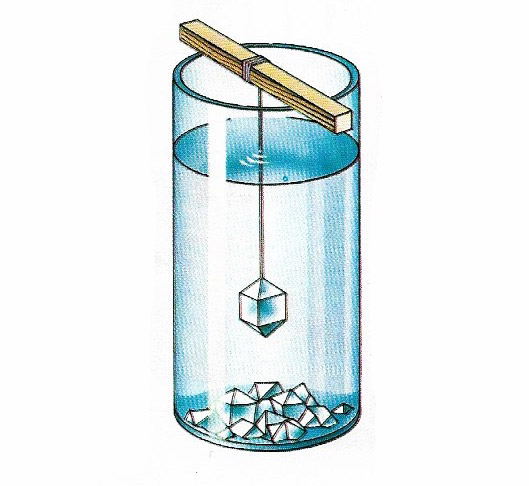
Crystals grow by precipitation out of a supersaturated solution or a cooling melt. The atoms or ions coalesce into tiny "seeds" around which further particles build up the lattice layers. If alum powder is dissolved in a little hot water with a drop of sulfuric acid and placed in a jar as shown, alum crystals will grow as the solution cools. Slower cooling gives larger crystals. Cooling molten sulfur causes the formation of sulfur crystals.
Crystallization is the process of forming crystals by a substance passing from a gas or liquid to the solid state (sublimation or fusion) or coming out of solution (precipitation or evaporation). In the fusion method a solid is melted by heating, and crystals form as the melt cools and solidifies. Ice crystals and monoclinic sulfur are formed in this way. Crystallization is an important laboratory and industrial technique for purifying and separating compounds.


