influenza
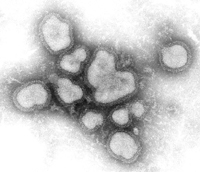
Influenza A virus. Credit: CDC.
Influenza is any of a group of viral diseases causing mild respiratory symptoms, fever, malaise, muscle pains, and headache, and often occurring in rapidly spreading epidemics. Gastrointestinal tract symptoms may also occur. Rarely, it may cause a severe viral pneumonia. The flu differs in several ways from the common cold. For example, people with colds rarely get fevers or headaches or suffer from the extreme exhaustion that flu viruses cause.
A characteristic of influenza (flu) viruses is their property of changing their antigenic (see antigen) nature frequently, so that immunity following a previous attack ceases to be effective.
Besides the rapid start of the outbreaks and the large numbers of people affected, the flu is an important disease because it can cause serious complications. Most people who get the flu get better within a week (although they may have a lingering cough and tire easily for a while longer). For elderly people, newborn babies, and people with certain chronic illnesses, however, the flu and its complications can be life-threatening.
Forms of flu
Flu is not the same as the common cold, nor is it related to what is commonly called the "stomach flu." Three different forms of flu are recognized.
Seasonal flu
Seasonal flu is the term used to refer to the flu outbreaks that occur yearly, mainly in the late fall and winter. It is estimated that between 5 and 20 percent of Americans come down with the flu every flu season.
Pandemic flu
Pandemic flu refers to particularly virulent strains of flu that spread rapidly from person to person to create a world-wide epidemic.
Bird (avian) flu
In nature, the flu virus also occurs in wild aquatic birds such as ducks and shore birds. It does not normally spread from birds to humans. However, pigs can be infected by bird influenza (as well as by the form of influenza that affects humans) and can pass on the flu to humans. In 1997, it was discovered that a virulent bird influenza had skipped the pig step and had infected humans directly, causing a number of deaths in Asia.
These instances of bird flu in humans have raised concerns that if this type of flu could at some point be transmitted between people, a new pandemic would occur. Thus, the term bird flu or avian flu is currently being used to refer to a possible pandemic flu.
The flu virus
A flu virus is roughly round, but it can also be elongated or irregularly shaped. Inside are eight segments of single-strand RNA containing the genetic instructions for making new copies of the virus. Flu's most striking feature is a layer of spikes projecting from its surface. There are two different types of spikes: one is the protein hemagglutinin (HA), which allows the virus to "stick" to a cell and initiate infection, the other is a protein called neuraminidase (NA), which enables newly formed viruses to exit the host cell.
Virus A, B, C
The first flu virus was identified in the 1930s. Influenza viruses are classified as type A, B, or C based upon their protein composition. Type A viruses are found in many kinds of animals, including ducks, chickens, pigs, whales, and also in humans. The type B virus widely circulates in humans. Type C has been found in humans, pigs, and dogs and causes mild respiratory infections, but does not spark epidemics.
Type A influenza is the most frightening of the three. It is believed responsible for the global outbreaks of 1918, 1957 and 1968. Type A viruses are subdivided into groups based on two surface proteins, HA and NA. Scientists have characterized 16 HA subtypes and 9 NA subtypes.
Naming viral strains
Type A subtypes are classified by a naming system that includes the place the strain was first found, a lab identification number, the year of discovery, and, in parentheses, the type of HA and NA it possesses, for example, A/Hong Kong/156/97 (H5N1). If the virus infects non-humans, the host species is included before the geographical site, as in A/Chicken/Hong Kong/G9/97 (H9N2). There are no type B or C subtypes.
Drifting and shifting
Influenza virus is one of the most changeable of viruses. These genetic changes may be small and continuous or large and abrupt.
Small, continuous changes happen in type A and type B influenza as the virus makes copies of itself. The process is called antigenic drift. The drifting is frequent enough to make the new strain of virus often unrecognizable to the human immune system. For this reason, a new flu vaccine must be produced each year to combat that year's prevalent strains.
Type A influenza also undergoes infrequent and sudden changes, called antigenic shift. Antigenic shift occurs when two different flu strains infect the same cell and exchange genetic material. The novel assortment of HA or NA proteins in a shifted virus creates a new influenza A subtype. Because people have little or no immunity to such a new subtype, their appearance tends to coincide with very severe flu epidemics or pandemics.
Transmission
You can get the flu if someone around you who has the flu coughs or sneezes. You can get the flu simply by touching a surface like a telephone or door knob that has been contaminated by a touch from someone who has the flu. The viruses can pass through the air and enter your body through your nose or mouth. If you've touched a contaminated surface, they can pass from your hand to your nose or mouth.
You are at greatest risk of getting infected in highly populated areas, such as in crowded living conditions and in schools.
Symptoms
If you get infected by the flu virus, you will usually feel symptoms 1 to 4 days later. You can spread the flu to others before your symptoms start and for another 3 to 4 days after your symptoms appear. The symptoms start very quickly and may include:
Typically, the fever begins to decline on the second or third day of the illness. The flu almost never causes symptoms in the stomach and intestines. The illness that some call "stomach flu" is not influenza.
Complications
You can have flu complications if you get a bacterial infection, which can cause pneumonia in your weakened lungs. Pneumonia also can be caused by the flu virus itself.
Complications usually appear after you start feeling better. After a brief period of improvement, you may suddenly get these symptoms
Pneumonia can be a very serious and sometimes life-threatening condition. If you have any of these symptoms, you should contact your health care provider immediately to get the appropriate treatment.
Flu complications in children and teenagers
Reye's syndrome, a condition that affects the nerves, sometimes develops in children and teenagers who are recovering from the flu. Reye's syndrome begins with nausea and vomiting, but the progressive mental changes (such as confusion or delirium) cause the greatest concern.
The syndrome often begins in young people after they take aspirin to get rid of fever or pain. Although very few children develop Reye's syndrome, you should consult a health care provider before giving aspirin or products that contain aspirin to children. Acetaminophen does not seem to be connected with Reye's syndrome.
Other complications of the flu that can affect children
Newborn babies recently out of intensive care units are particularly vulnerable to suffering from flu complications.
Treatment
If you do get the flu and want to take medicine to treat it, your health care provider may prescribe one of four available antiviral medicines.
Flu drugs
To work well, you must take these medicines within 48 hours after the flu begins. They reduce the length or time fever and other symptoms last and allow you to more quickly return to your daily routine.
How flu drugs work
In 1976, the Food and Drug Administration (FDA) approved amantadine to both treat and prevent influenza type A in adults and children 1 year old or older. FDA approved rimantadine – a derivative of amantadine – in 1993 to treat and prevent influenza infection in adults and prevent influenza in children.
These two drugs act against influenza A viruses but not against influenza B viruses. These compounds inhibit the activity of the influenza virus M2 protein, which forms a channel in the virus membrane. As a result, the virus cannot replicate after it enters a cell. The drug manufacturers recommend daily doses for using amantadine and rimantadine to treat and prevent the flu in different age groups. Researchers, however, have not adequately evaluated amantadine and rimantadine in children younger than 1 year old.
In 1999, FDA approved two additional drugs to fight the flu: zanamivir and oseltamivir, the first of a new class of antiviral drugs called neuraminidase inhibitors.
The surfaces of influenza viruses are dotted with neuraminidase proteins. Neuraminidase, an enzyme, breaks the bonds that hold new virus particles to the outside of an infected cell. Once the enzyme breaks these bonds, this sets free new viruses that can infect other cells and spread infection. Neuraminidase inhibitors block the enzyme's activity and prevent new virus particles from being released, thereby limiting the spread of infection.
Are these drugs effective against any kind of influenza virus?
Rimantadine and amantadine are effective only against type A influenza. Zanamivir and oseltamivir inhibit both influenza A and B viruses.
Who should consider using these drugs?
Amantadine is approved for treating and preventing uncomplicated influenza A virus infection in children who are 1 year of age or older and adults. Rimantadine is approved for treating and preventing uncomplicated influenza virus A infection in adults and for preventing, but not treating, such infections in children.
Zanamivir is approved for preventing influenza A and B virus infections in people 5 years of age and older and for treating uncomplicated influenza virus infection in people 7 years of age and older who have not had symptoms for more than 2 days.
Oseltamivir is approved for treating uncomplicated influenza virus infection in people 1 year of age or older who have not had symptoms for more than 2 days. Oseltamivir is available in a pediatric liquid formulation. Oseltamivir also is approved for preventing influenza A and B in people 1 year of age and older.
How are these drugs administered?
How much do the drugs help?
Studies have shown that all four drugs can reduce the duration of flu symptoms by 1 day if taken within 2 days of the onset of the illness. There is no information about how effective these drugs are if treatment is started more than 2 days after onset of flu symptoms.
When taken as directed to prevent the flu, oseltamivir can significantly reduce your chance of getting the disease if there is a flu outbreak in your family or community.
Amantadine and rimantadine have been reported to prevent the spread of influenza A outbreaks primarily in nursing homes. If someone in your family is diagnosed with influenza, taking one of these drugs may reduce your chances of getting the disease.
Other benefits
None of the four drugs has been shown to effectively prevent serious influenza-related complications such as bacterial or viral pneumonia. Studies of the use of zanamivir in families and in nursing homes at risk for influenza infection, however, resulted in the reduced use of antibiotics, which are frequently prescribed to treat these serious complications.
Side effects
The neuraminidase inhibitors generally cause fewer side effects than the older flu drugs.
The most common side effects seen with oseltamivir are nausea and vomiting. Some adverse reactions have been reported in children. In some people, zanamivir can cause wheezing or serious breathing problems that need prompt treatment. The other most common side effects seen with zanamivir include headache and diarrhea.
Amantadine and rimantadine can cause side effects such as insomnia and anxiety, nausea or loss of appetite. In some cases, severe side effects such as seizures have been reported.
Should certain people not take these drugs?
People allergic to these drugs or their ingredients should not take them.
Zanamivir generally is not recommended for people with chronic respiratory diseases such as asthma or chronic obstructive pulmonary disease (COPD). In clinical studies, some patients with mild or moderate asthma or COPD had bronchospasm (wheezing) after taking zanamivir. If you have an underlying respiratory disease and have been prescribed zanamivir, your health care provider should instruct you to have a fast-acting inhaled bronchodilator available for use when taking zanamivir.
The dosage of oseltamivir may need to be adjusted if you have any type of kidney disease.
None of these drugs is recommended for routine use during pregnancy or nursing. These drugs have not been evaluated in pregnant women, and researchers do not know the effects these drugs could have on the unborn child.
In laboratory and in limited clinical studies, there have been no reported interactions of the neuraminidase inhibitors with other drugs.
Can influenza viruses develop resistance to these drugs?
When either amantadine or rimantadine is used for therapy, drug-resistant flu viruses may appear in about one-third of patients.
Laboratory studies have shown that influenza A and B viruses can develop resistance to zanamivir and oseltamivir. Surveillance for neuraminidase inhibitor-resistance has been started.
How do the flu drugs compare?
No study to date has directly compared the effectiveness of zanamivir, oseltamivir, amantadine, and rimantadine for treating influenza A or zanamivir and oseltamivir for treating influenza A or B. The available information suggests that these four drugs are similarly effective in reducing the duration of uncomplicated acute illness due to influenza A.
Zanamivir and oseltamivir differ from amantadine and rimantadine in terms of their side effects and cost. Use of amantadine, and to a lesser extent rimantadine, has been associated with central nervous system side effects including nervousness, anxiety, insomnia, and light-headedness. These side effects have not been associated with zanamivir or oseltamivir. On the other hand, zanamivir and oseltamivir are significantly more expensive than either rimantadine or amantadine. All four drugs are available by prescription only, and it is best to consult with your health care provider to determine what drug might be best for you.
Prevention
Flu vaccine
The main way to keep from getting flu is to get a yearly flu vaccine. You can get the vaccine at your doctor's office or a local clinic, and in many communities at workplaces, supermarkets, and drugstores. You must get the vaccine every year because it changes.
Scientists make a different vaccine every year because the strains of flu viruses change from year to year. Nine to 10 months before the flu season begins, they prepare a new vaccine made from inactivated (killed) flu viruses. Because the viruses have been killed, they cannot cause infection. The vaccine preparation is based on the strains of the flu viruses that are in circulation at the time. It includes those A and B viruses (see section below on types of flu viruses) expected to circulate the following winter.
Sometimes, an unpredicted new strain may appear after the vaccine has been made and distributed. Because of this, even if you do get the flu vaccine, you still may get infected. If you do get infected, however, the disease usually is milder because the vaccine will still give you some protection.
Until recently, you could get the flu vaccine only as an injection. In 2003, however, the FDA approved a nasal spray flu vaccine called FluMist that you can get from your health care provider. The FDA approved it for use in healthy people aged 5 to 49 years. You should not use FluMist if
Children or teenagers who regularly take aspirin or products containing aspirin also should not take FluMist.
Your immune system takes time to respond to the flu vaccine. Therefore, you should get vaccinated 6 to 8 weeks before flu season begins in November to prevent getting infected or reduce the severity of flu if you do get it. Because the flu season usually lasts until March, however, it's not too late to get it after the season has begun. The vaccine itself cannot cause the flu, but you could become exposed to the virus by someone else and get infected soon after you are vaccinated.
Side effects
You should be aware that the flu vaccine can cause side effects. The most common side effect in children and adults is soreness at the site of the vaccination. Other side effects, especially in children who previously have not been exposed to the flu virus, include fever, tiredness, and sore muscles. These side effects may begin 6 to 12 hours after vaccination and may last for up to 2 days.
Viruses for producing the vaccine are grown in chicken eggs and then killed with a chemical so that they can no longer cause an infection. The flu vaccine may contain some egg protein, which can cause an allergic reaction. Therefore, if you are allergic to eggs or have ever had a serious allergic reaction to the flu vaccine, The Centers for Disease Control and Prevention (CDC) recommends that you consult with your health care provider before getting vaccinated.
Vaccine recommendations
If you are in any of the following groups or live in a household with someone who is, CDC recommends that you get the flu vaccine.
CDC recommends that children 6 months to 23 months of age get the flu vaccine.
Children and teenagers (6 months to 18 years of age) should get the flu vaccine if they are taking long-term aspirin treatment as they may be at risk of developing Reye's syndrome following a flu infection (see section on complications in children). They should also get the flu vaccine if they live in a household with someone in the above groups.
Health care providers and volunteers should get the flu vaccine if they work with people in any of the above groups.
Medicine for prevention
Although the flu vaccine is the best way to prevent getting the flu, four antiviral medicines also are available by prescription that will help prevent
Tamiflu, Flumadine, and Symmetrel may be used by children who are 1 year of age and older and adults. For adults and children 5 years of age and older, Relenza is used for prevention of flu.


