methanal
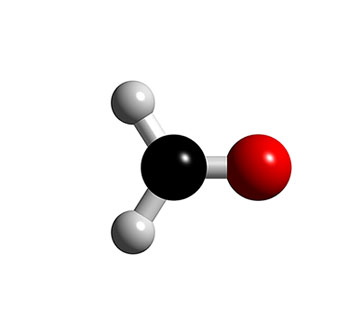
Methanal (HCHO) is a colorless, acidic, toxic gas, also known as formaldehyde. Methanal is the simplest and most reactive of the aldehydes, and the aldehyde derivative of formic acid. It was discovered in 1867 by Augustus von Hofmann.
Methanal is made by catalytic air oxidation of methanol vapor or of natural gas. Methanal gas is unstable and is usually stored as its aqueous solution (35–40% methanal), formalin, which is used as a disinfectant and preservative for biological specimens; on keeping, formalin deposits a polymer, polyoxymethylene (paraformaldehyde), which regenerates methanal on heating.
Methanal is condensed with urea and phenols to make plastics, with ammonia to give hexamethylenetetramine (a urinary antiseptic also used to make RDX), and with ethanal (acetaldehyde) to give penaerithrytol and hence PETN. It is also used in tanning and textile manufacture.
| relative molecular mass | 30.0 |
| density relative to water | 0.82 (at -20°C) |
| melting point | -92°C (-133.6°F) |
| boiling point | -19°C (-2.2°F) |


