band spectrum
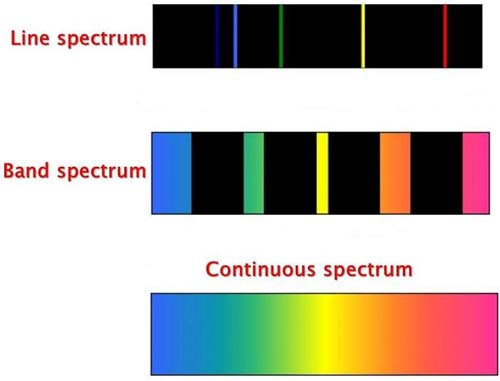
A band spectrum is a molecular spectrum consisting of numerous closely spaced, often unresolved, emission lines or absorption lines which occur across a limited range of frequencies. Each line represents an increment of energy due to a change in the rotational state of a specific molecule.
The study of band spectra has played a crucial role in our understanding of the electronic structure of atoms and molecules, as well as in the development of technologies such as lasers and solar cells.
At its most basic, a band spectrum arises when a substance absorbs or emits light at specific wavelengths or frequencies. These wavelengths or frequencies correspond to the energy levels that are present in the substance, and the intensity of the absorption or emission is proportional to the number of atoms or molecules that are present at each energy level.
The energy levels in a substance are typically organized into "bands," which are characterized by a range of energy levels that are closely spaced together.
There are two main types of band spectra: absorption spectra and emission spectra. An absorption spectrum is produced when a substance absorbs light, and is typically measured by shining light on the substance and measuring the amount of light that is absorbed at each wavelength or frequency. An emission spectrum, on the other hand, is produced when a substance emits light, and is typically measured by observing the light that is emitted by the substance when it is excited by an external energy source.
One of the key features of band spectra is that they are highly characteristic of the substance being studied. Different substances have different arrangements of energy levels, and as a result, they have distinct band spectra. This property of band spectra has made them a powerful tool for identifying and characterizing unknown substances, as the unique pattern of bands in a substance's spectrum can often be used to identify it.
In addition to their use in identification and characterization, band spectra have also played a key role in our understanding of the electronic structure of atoms and molecules. By studying the energy levels in a substance, scientists can gain insights into the arrangement of electrons in the substance, as well as the forces that hold these electrons in place. This understanding has been crucial in the development of technologies such as lasers, which rely on the ability to control the movement and arrangement of electrons in order to produce coherent light.
Band spectra have also found a variety of other applications, including in the development of solar cells and in the study of materials science. In solar cells, band spectra can be used to understand the efficiency with which a given material absorbs light and converts it into electrical energy. In materials science, band spectra can be used to study the properties of materials and to understand how they respond to different stimuli, such as pressure or temperature.
Bands caused by titanium oxide, zirconium, and carbon compounds are characteristic of low temperature stars.


