sandwich compound
A sandwich compound is an organometallic compound in which the metal atom is sandwiched between two aromatic-ringed ligands – the whole of the ring electron system interacting with the metal orbitals, giving great stability. Ferrocene and dibenzene-chromium are examples.
Ferrocene
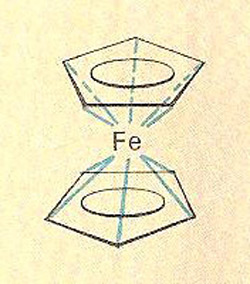 |
| Ferrocene.
|
Ferrocene, also called dicyclopentadienyl iron [(C5H5)2Fe), is an orange crystalline organo-iron compound. It is a typical, stable sandwich compound – the first to be prepared (1951) – consisting of an iron (II) ion sandwiched between two five-member carbon rings. The molecular structure is a pentagonal antiprism. Melting point 174°C, sublimation 100°C.


