X-ray diffraction
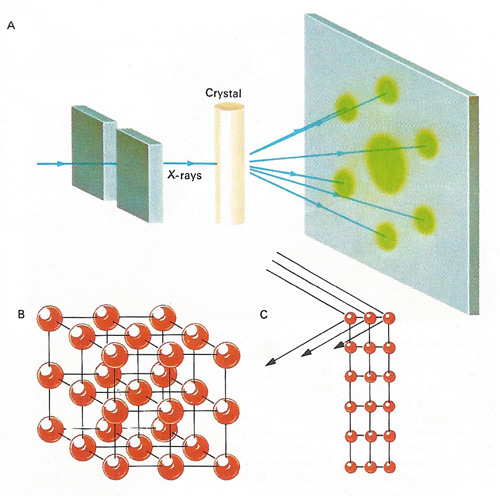
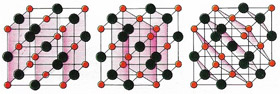
X-rays diffract from the periodic regular array of unit cells in a crystal to produce a pattern of diffraction spots [A]. X-ray waves reflecting from successive planes need to be a whole number of wavelengths out of step in order to produce a diffraction spot [C]. Symmetries and regularities in the pattern help the crystallographer to decide which type of unit cell is present. For instance the diffraction pattern in [A] shows hexagonal symmetry, so a unit cell such as those in [B] cannot be present. From the relative intensities of the spots and their phases one can calculate the crystal structure.
X-ray diffraction is the diffraction of X-rays by the atoms or ions of a crystal, according to a characteristic pattern that enables information to be obtained on the structure of the crystal. X-ray diffraction was discovered by Max von Laue in 1912 and developed for crystal analysis by William Henry and William Lawrence Bragg in 1912–13.
The atomic nuclei in a crystal lattice act as diffraction gratings; the planes of atoms have spacings of a few angstrom units, which are comparable with the wavelengths of X-rays. Strong scattering of the rays by the crystal therefore occurs in certain directions, according to Bragg's law. Various techniques are available for applying X-ray diffraction to the study of single crystals, powders, fibers, etc., and by computation, using Fourier synthesis, it is possible to work out three-dimensional electron density maps of the solid lattice from the recorded X-ray patterns.
 |
| X-rays are scattered in various directions by crystal lattices, depending on the distance between the lattice planes. In diffraction apparatus (A), the source (1) generates single-wavelength X-rays to hit an oriented crystal (2) on a revolving support (3). Diffraction occurs as the beam passes between the lattice planes (B) in accordance with Bragg's law. The diffracted rays are in phase at certain angles, producing rows of spots on a cylindrical photographic film (4) or curved lines of spots on a flat plate (C). Different planes (D) with different orientations may exist within the same crystal. |


