forbidden line
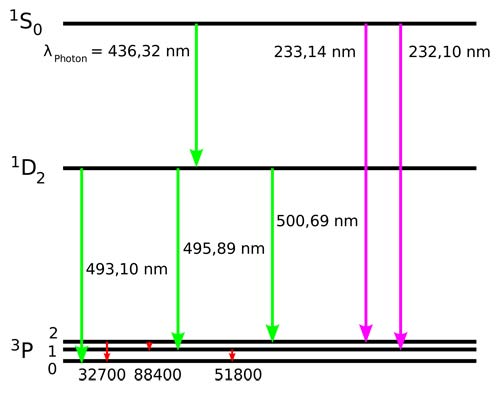
Electron transitions of doubly ionized oxygen: forbidden transitions in the visible spectrum are shown in green.
A forbidden line is an emission line found in the spectrum of a rarefied gas under special conditions, such as those found in some nebulae, the solar corona, and parts of active galactic nuclei. Although not seen on Earth – hence their name – forbidden lines may account for 90% or more of the total visual brightness of an object such as a planetary nebula.
A forbidden line arises when an electron in an excited (energized) atom jumps from a metastable state to a lower energy level. Under normal circumstances, when particle densities are higher (greater than about 108 per cubic centimeter), such an electron would almost immediately be knocked out of its metastable state by collision and not be given time to emit a photon. But in an environment like that of a planetary nebula, the time between collisions averages 10 to 10,000 seconds. Consequently, when ions such as O+, O2+ (singly and doubly ionized oxygen), or N+ (singly ionized nitrogen) go into metastable states by allowed transitions from higher states, they remain there undisturbed until they radiate spontaneously. A large fraction of the more highly excited ions eventually drop into these states and, in a nebular environment, practically every ion goes from them to the ground state by forbidden radiation.
Forbidden lines are denoted by enclosing them in brackets. The strongest are two lines of doubly ionized oxygen [O III], in the green part of the spectrum (to which the human eye is most sensitive) at 4959 and 5007 Å. When these lines were first seen, in the spectra of planetary nebula in the 1860s, their true nature wasn't recognized and it was thought they might be due to a new element, which was dubbed "nebulium." More than half a century passed before Ira Bowen provided the right explanation. Besides forbidden lines of ionized oxygen, others of neon, nitrogen, and other relatively abundant elements are seen making up the light of nebulae, as well as the ordinary, permitted lines of hydrogen and helium.


