carbide
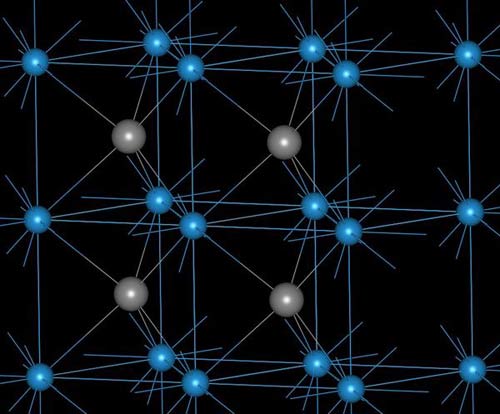
Alpha tungsten carbide crystal structure.
A carbide, also called an acetylide, is an inorganic compound of carbon with other metals or other more electropositive elements. Boron and silicon form extremely hard carbides which are used as abrasives. Many transition elements also form carbides, in which carbon atoms occupy spaces between adjacent atoms in the metal lattice. Some electropositive metals form ionic carbon compounds; the best is calcium carbide (CaC2), which reacts with water to give acetylene (ethyne).


