cysteine
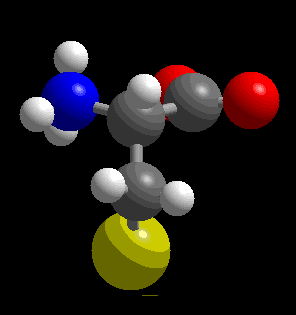
Ball-and-stick representation of the cysteine molecule. Gray=C, red=O, blue=N, yellow=S, white=H.
Cysteine is a sulfur-containing amino acid (HSCH2CH2COOH) that occurs in animal proteins, especially those in hair, hooves, and the keratin of skin; the HS– group in it makes it the catalytic element in some enzymes. The disulfide (S-S) links between adjacent cysteine molecules in polypeptide chains contribute to the three-dimensional molecular structure of proteins.
Acetylcysteine
Acetylcysteine, also known as N-acetylcysteine, is a drug administered as eye drops to break down mucus filaments in the eyes in the treatment of dry eyes (for example, in Sjögren's syndrome). Acetylcysteine is also given, by intravenous infusion, to prevent liver damage in paracetamol overdose. Trade names: Acetadote (injection), Ilube, Mucomyst.
Homocysteine
Homocysteine is an intermediate in the synthesis of cysteine. A deficiency in the enzyme cystathionine synthetase results in elevated levels in the blood of homocysteine and homocystine (an oxidized form of homocysteine), resulting in elevated urinary levels (homocystinuria). It is becoming increasingly recognized that elevated levels of homocysteine in the blood are a risk factor for vascular disease independent of diabetes, hypertension, elevated levels of cholesterol in the blood, and smoking. Levels are also elevated in vitamin B12 deficiency.


