noble gas

Noble gas discharge tubes. Image copyright: smart-elements.com.
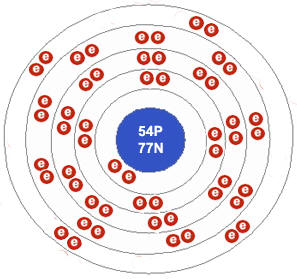
A xenon atom. Notice that the outer electron shell has 8 electrons and is therefore full. This makes xenon extremely unreactive.
A noble gas is any of a family of (almost) completely unreactive, colorless, and odorless elements. The noble gases are (in order of increasing atomic number):
• helium
• neon
• argon
• krypton
• xenon
• radon
The noble gases occupy group 0 (or group 18, depending on the scheme adopted) of the periodic table. Their extremely low reactivity is due to the fact that their outer electron shells are complete (having two electrons in the case of helium and eight electrons in the case of the other inert elements). Being already complete, the electron shells of noble gases "seek" neither to lose electrons or gain them during chemical reactions and thus tend not to react at all.
It was thought formerly that all of these elements were completely inert. But it's now known that argon, krypton, xenon, and radon do form compounds (most notably fluorides) under certain circumstances. The noble gases are also called inert gases or rare gases, although argon and helium occur in substantial quantities in Earth's atmosphere (one percent in the case of argon). They are prepared by fractional distillation of liquid air, except helium and radon.
Discharges of the noble gases
The noble gases glow brightly when an electric discharge is passed through them, and so are used as advertising signs: neon tubes glow red, xenon blue, and krypton bluish-white; argon tubes glow pale red at low pressures, blue at high pressures.


